 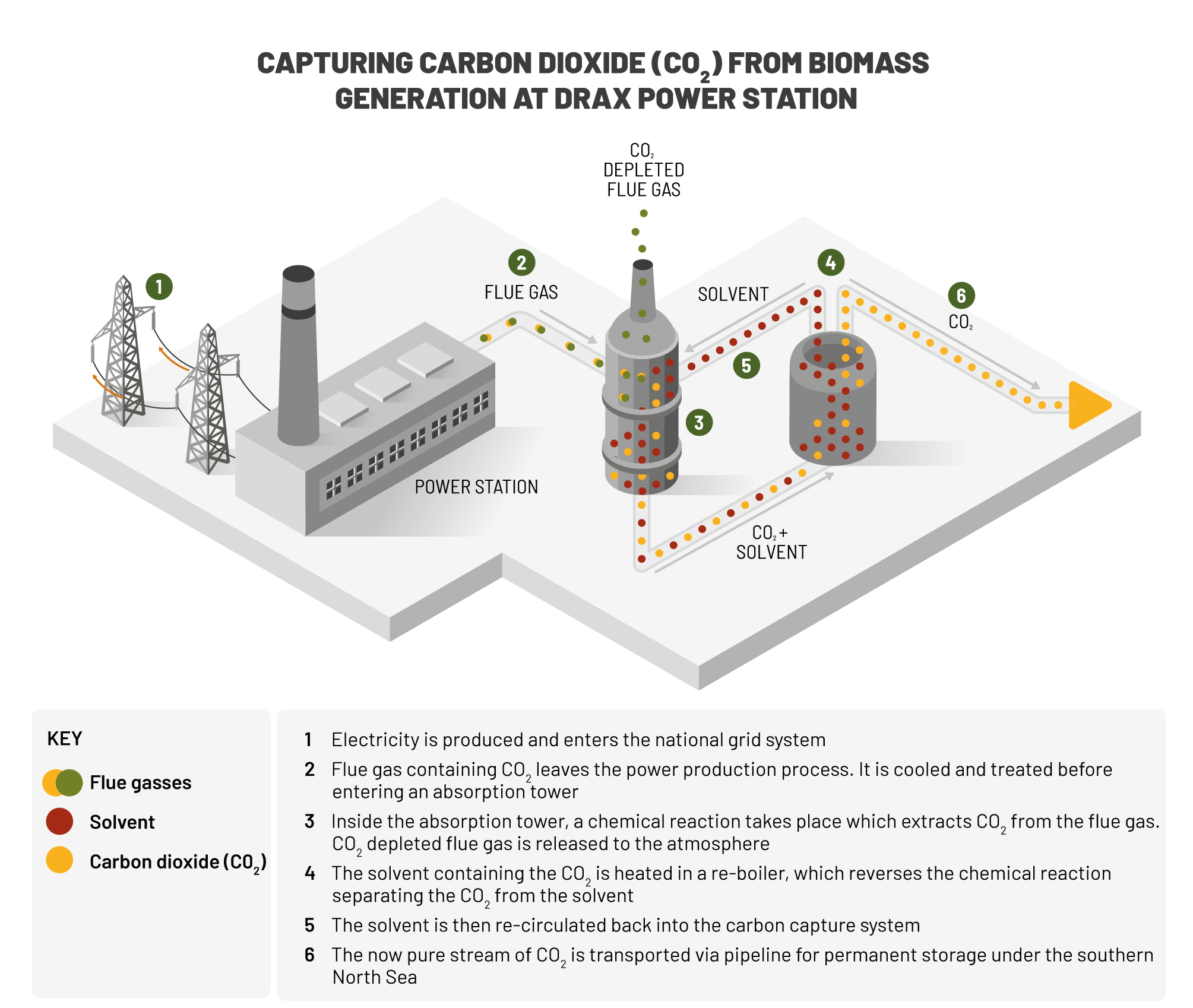 
This releases the trapped CO2, which can then be transported for storage elsewhere. In the most commonly-used techniques, once the chemicals in the absorber column become saturated, a stream of superheated steam at around 120C is passed through it. Post-combustion In the post-combustion method, CO2 is separated from the flue gas of the power station by bubbling the gas through an absorber column packed with liquid solvents (such as ammonia) that preferentially take out the CO2. The pilot is an oxyfuel boiler that can generate 30MW of heat and around 12MW of electricity. But, so far, only a small pilot project at Vatenfall's Schwarze Pumpe power station in northern Germany has connected all the different stages of the CCS chain together. The oldest power stations may end up being uneconomical to refit.Īll the components of CCS have been in use by oil companies and chemical engineering plants for decades. Pipes to transport the CO2 to suitable burial sites could cost £1m per mile. The technology is not cheap – up to 40% of a power station's energy could end up being used to run the CCS scrubbing and transport systems and experts estimate the average cost of retro-fitting Britain's aged power stations at about £1bn each. The last method is often used in a process called "enhanced oil recovery", where CO2 is pumped into an oil field to force out the remaining pockets of oil that would otherwise prove difficult to extract. Once the CO2 has been trapped, it is liquefied, transported – sometimes for several hundred miles – and buried, either in suitable geological formations, deep underground saline aquifers or disused oil fields. The third method, oxyfuel, burns the coal in an atmosphere with a higher concentration of pure oxygen, resulting in an exhaust gas that is almost pure CO2. Pre-combustion CCS takes place before the fuel is placed in the furnace by first converting coal into a clean-burning gas and stripping out the CO2 released by the process. There are three main techniques: the post-combustion process involves scrubbing the power plant's exhaust gas using chemicals. The carbon dioxide reacts with the calcium hydroxide to form white calcium carbonate, which is insoluble and so turns the limewater ‘milky’.Ĭalcium hydroxide + carbon dioxide → calcium carbonate + waterĬa(OH) 2 (aq) + CO 2 (g) → CaCO 3 (s) + H 2 O(l)Īll of these reactions are linked together and form the limestone cycle. Ĭalcium hydroxide dissolves in excess water to produce calcium hydroxide solution ( limewater ), which is used to test for carbon dioxide. The solid remains white but crumbles into a powder as the water is added.Ĭalcium oxide + water → calcium hydroxideĬaO(s) + H 2 O(l) → Ca(OH) 2 (s) Calcium hydroxideĬalcium hydroxide (also known as slaked lime ) is used to neutralise excess acidity, for example, in lakes and soils affected by acid rain. This is an exothermic reaction, indicated by the water turning to steam. Calcium oxideĬalcium oxide reacts with a few drops of water to form calcium hydroxide, which is an alkali. This is indicated by an orange glow as the limestone is heated.Ĭalcium carbonate → calcium oxide + carbon dioxideĬalcium oxide (also known as quicklime ) is a key ingredient in the making of cement and is also used to make certain types of plaster. When limestone is heated strongly, the calcium carbonate it contains absorbs heat ( endothermic ) and decomposes to form calcium oxide. Calcium carbonate, calcium oxide and calcium hydroxide are all made from limestone and have important applications so it is important to know how they are made.Ĭalcium carbonate is found naturally in limestone. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
